

Vol. 11, Issue 3

Managing the Dose:
The VASP Assay for Anti-P2Y12 Drugs
Originally published in Drug Discovery & Development, February, 2011, p. 24. See original article here
In a clinical trial, the selection of an appropriate dose of a drug leads to optimal efficacy and safety as the trial progresses. By necessity, assays are developed that specifically measure responsiveness to a drug. Case-in-point, management of anti-clotting drugs has recently received significant attention from the pharmaceutical industry, cardiologists, and drug regulators alike, due to the fact that these drugs have been associated with bleeding following injury or surgery.
 Thienopyridines are one class of drugs with anti-clotting properties; the most well-known is clopidogrel (Plavix, Pfizer). By competing for binding with the platelet agonist ADP, clopidogrel inhibits clotting by directly interfering with normal platelet cell signaling after binding to the ADP/P2Y12 platelet cell surface receptor. Binding of ADP leads to normal signaling through the ADP/P2Y12 receptor, which ultimately leads to the involvement of a phosphoprotein known as vasodilator-stimulated phosphoprotein (VASP). By triggering prostaglandin E1 (PGE1) receptor-mediated signaling, binding of (PGE1) alone leads to phosphorylation of VASP and platelet inactivation, while incubating platelets with both ADP and PGE1 has the opposite effect. In contrast, when platelets are first treated with a P2Y12 antagonist (such as clopidogrel) and then incubated with PGE1 either alone or in combination with ADP, VASP is constitutively phosphorylated and the platelets do not form a clot.
Thienopyridines are one class of drugs with anti-clotting properties; the most well-known is clopidogrel (Plavix, Pfizer). By competing for binding with the platelet agonist ADP, clopidogrel inhibits clotting by directly interfering with normal platelet cell signaling after binding to the ADP/P2Y12 platelet cell surface receptor. Binding of ADP leads to normal signaling through the ADP/P2Y12 receptor, which ultimately leads to the involvement of a phosphoprotein known as vasodilator-stimulated phosphoprotein (VASP). By triggering prostaglandin E1 (PGE1) receptor-mediated signaling, binding of (PGE1) alone leads to phosphorylation of VASP and platelet inactivation, while incubating platelets with both ADP and PGE1 has the opposite effect. In contrast, when platelets are first treated with a P2Y12 antagonist (such as clopidogrel) and then incubated with PGE1 either alone or in combination with ADP, VASP is constitutively phosphorylated and the platelets do not form a clot.
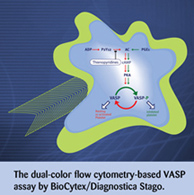
Based on its differential phosphorylation profile in the presence of an anti-P2Y12 drug, VASP is the most reliable molecular reporter of platelet function. Platelet responsiveness to anti-P2Y12 drugs can be measured by a number of different platelet function assays, but only one assay directly measures VASP phosphorylation: The dual-color flow cytometry-based VASP assay developed by BioCytex (Marseille, France) and Diagnostica Stago.
The assay workflow begins with an uncoagulated whole blood specimen collected on citrate from patients who are currently taking an anti-P2Y12 drug. A 30-microliter sample of specimen is incubated with PGE1 in the presence and absence of ADP. This initial incubation period is immediately followed by fixation and permeabilization. Fixed, permeabilized cells are first labeled with a primary antibody against the phosphorylated Ser-239 residue of VASP (mouse monoclonal anti-VASP-P). This is followed by staining with fluorescein isothiocyanate (FITC)-conjugated goat anti-mouse polyclonal antibody and counterstaining with antibody against platelet surface marker CD61. Background fluorescence is assessed by simultaneously running a negative isotype control. The VASP assay protocol can be completed in 30 minutes at room temperature. Flow cytometric analysis of sample fluorescence generates a statistic known as the Platelet Reactivity Index (PRI), which is an estimate of the level of responsiveness to the anti-P2Y12 drug tested.
Several published clinical trials have demonstrated the usefulness of the VASP assay for the direct measurement of patient responsiveness to anti-P2Y12 drugs.1-3 Managing the dosage of anti-P2Y12 drugs is necessary to ensure their safety and efficacy during clinical development. The VASP assay enables drug researchers to assess patient responsiveness at specific dosages and better manage dose.
The VASP assay is labeled for research use only; not for use in diagnostic procedures in the United States & Canada.
For additional information, contact us at 1-800-222-COAG (2624) or customer.care@stago-us.com
Five Century Drive Parsippany, NJ 07054
1-800-222-COAG
973-631-1200
Fax: 973-631-1618 www.stago-us.com

References:
- Pampuch A, Chiara C, Giovanni de Gaetano. Thromb Haemost. 2006;96:767.
- Angiolillo DJ, Suryadevara S. Best Practice & Research Clinical Endocrinology & Metabolism. 2009;23:375.
- Gurbel PA, et al. Circulation. 2010;121:1188.